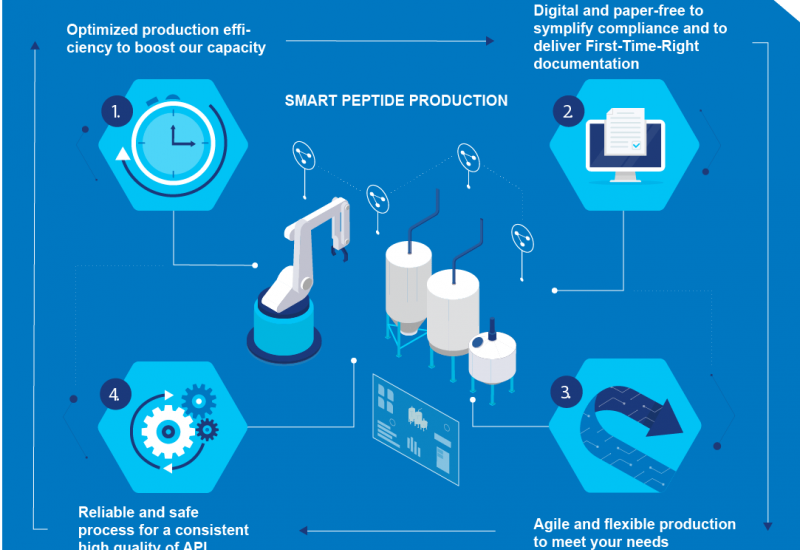
New LPPS technology from Cambrex
Cambrex’s Snapdragon Chemistry subsidiary has developed a new liquid-phase peptide synthesis (LPPS) technology that uses traditional API batch reactors and continuous flow, obviating the dependency on specialised, solid-phase reactors. It also “materially reduces solvent demand and the need for excess reagents compared to standard solid-state peptide synthesis (SPPS) processes”.